- Services
- Electrocardiogram (ECG) Testing
- 2D Echocardiogram (Heart Ultrasound)
- Treadmill Stress Test (TMT)
- Clinical Laboratory Services
- 24×7 On-Site Pharmacy
- Coronary Angiogram & Advanced Angiography
- Coronary Angioplasty & Stenting
- Intravascular Lithotripsy (IVL) & IVUS/OCT Imaging
- Pacemaker & ICD Implantation
- CRT-P/CRT-D Therapy (Cardiac Resynchronization)
- Pericardiocentesis (Fluid Drainage)
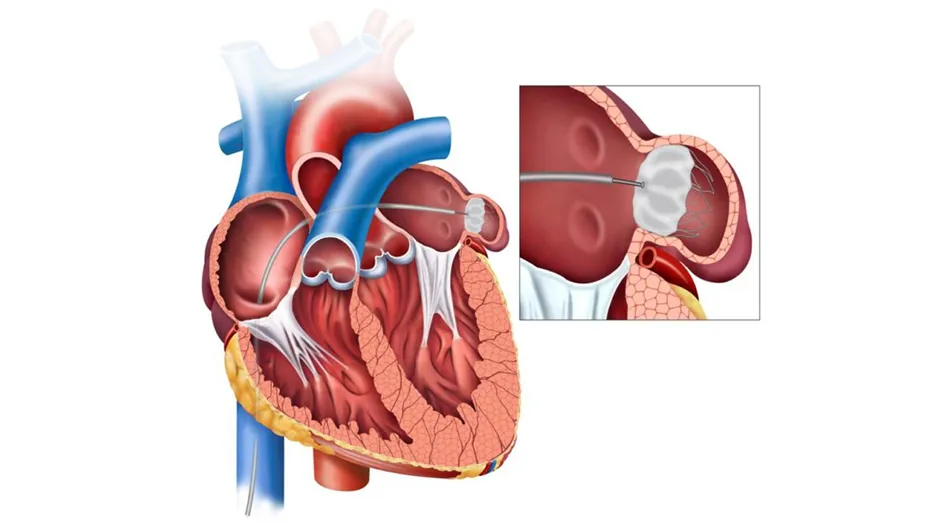
- Balloon Mitral Valvotomy
- ASD, VSD & PDA Device Closure
- Transcatheter Aortic Valve Replacement (TAVR)
- 24-Hour Holter Monitoring
- Ambulatory Blood Pressure Monitoring
- Sleep Study & Sleep Apnea Screening
- Peripheral Angioplasty & Vascular Interventions
- About Us
- Contact
- Privacy Policy
- Terms and Conditions
Shopping cart
At Pride Heart Institute, we combine cutting-edge technology with compassionate care to deliver excellence in cardiac health, community wellness, and lifelong support.
Sangareddy, Telangana 502001
Contact Us
+91 98668 74777
Monday - Sunday